SmofKabiven Central, SmofKabiven Electrolyte Free Central and SmofKabiven Peripheral offer a well-balanced combination of nutrients that can help meet the nutritional needs of hospitalised adult patients, including those with fluid restriction.1–3 These products contain SMOFlipid®4 (soya-bean oil, medium-chain triglycerides, olive oil and fish oil), which has been shown to have less effect on liver function and to reduce hospital length of stay compared with older lipid emulsions in PN regimens.5–7
For total PN, vitamins and trace elements should be administered to the patient in addition to the macronutrients delivered via multi-chamber bags.
Figures are approximations calculated from the summaries of product characteristics.

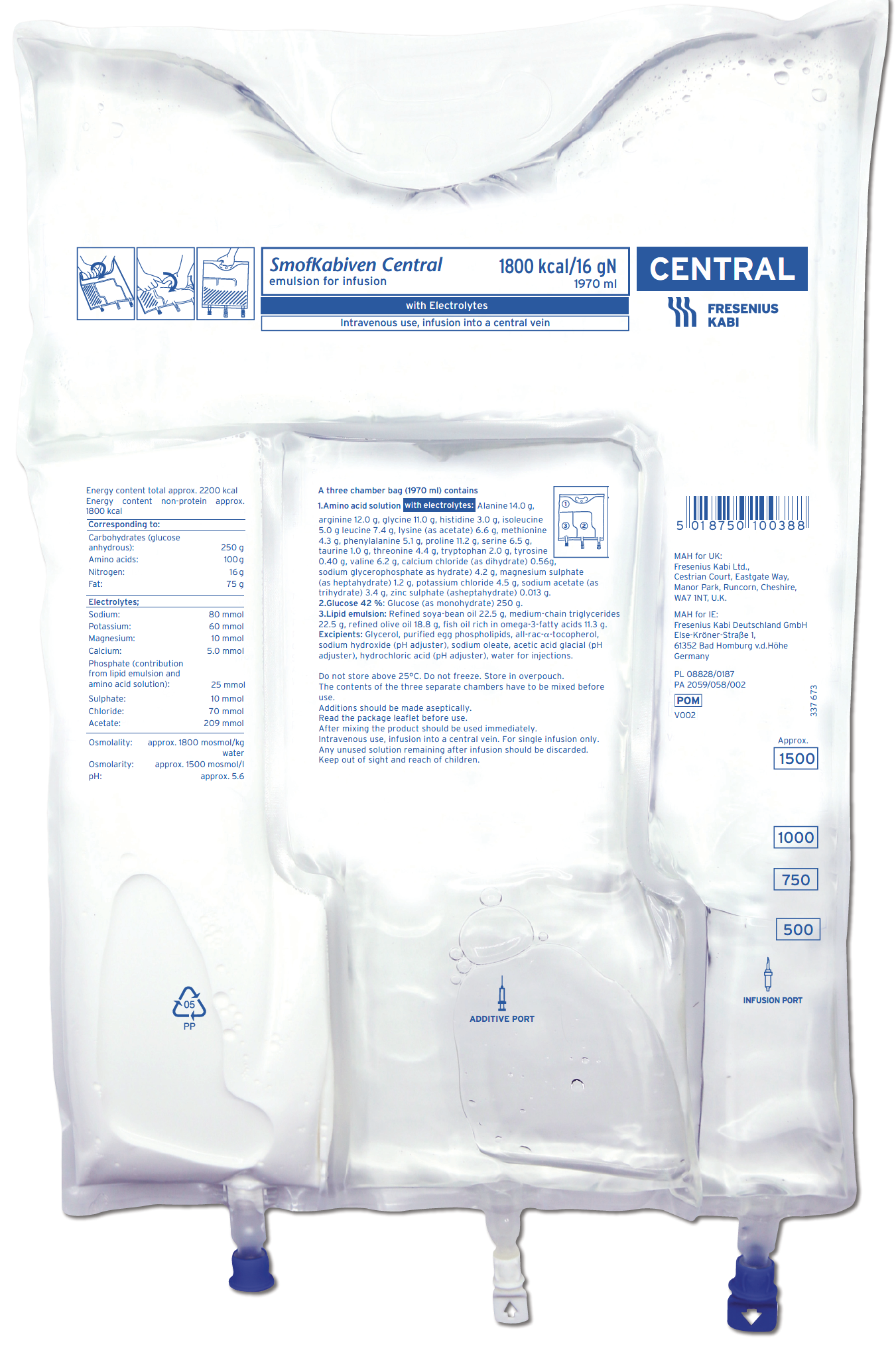
SmofKabiven Central1,2
SmofKabiven Peripheral3
Contains SMOFlipid a four lipid emulsion
Comprises 30% soya-bean oil providing omega-6 fatty acids, 30% medium-chain triglycerides providing rapidly available energy, 25% olive oil providing omega-9 fatty acids and 15% fish oil providing omega-3 fatty acids4
Omega-3 fatty acids have proven benefits for critically ill patients.8,9 * Compared with standard ** PN, hospitalised adults who received omega 3 fatty-acid enriched PN had a:9
- 40% lower risk of infection (131 vs 215 events; RR 0.60, 95% CI 0.49–0.72; p<0.00001) – primary endpoint
- 30-day mortality rate (83 vs 101 events; RR 0.84, 95% CI 0.65–1.07; p=NS)
- 56% lower risk of sepsis (24 vs 54 events; RR 0.44, 95% CI 0.28–0.70; p=0.0004)
- Shorter ICU stay (mean stay length reduced by 1.95 days, 95% CI 0.42–3.49; p=0.01)
- Shorter hospital stay (mean stay length reduced by 2.14 days, 95% CI 1.36–2.93; p<0.00001)
Appropriate volume
SmofKabiven Central provides calories, amino acids and a four oil lipid emulsion with purified fish oil, all within an appropriate volume
- Delivers finely balanced nutrition which can help achieve nutritional targets recommended in international guidelines (with or without electrolytes), including lower volume products for fluid restricted patients10–14
Balanced protein–energy ratio
SmofKabiven Central
Protein – energy ratio: 1.2 gAA/25 kcal
Nitrogen (g) – non-protein kcal ratio: 1:108
SmofKabiven Peripheral
Protein – energy ratio: 1.3gAA/28kcal
Nitrogen (g) – non-protein kcal ratio: 1:110
- Offers a nitrogen: non-protein–energy ratio aimed at meeting the nutritional requirements of the majority of adult patients requiring PN12–17
- Provides a ratio of calories and proteins to help to avoid metabolic complications in patients receiving PN13–16
Concentrated amino acids
SmofKabiven Central provides a concentrated amount (total amino acids 51 g/1000ml) of essential, non-essential and conditionally essential amino acids1,2
Contains taurine which may have a hepatoprotective effect1,2,18
SmofKabiven Central is available with or without electrolytes1,2
- SmofKabiven Central
- SmofKabiven Electrolyte Free Central
SmofKabiven Central is available in 4 sizes.1
SmofKabiven Electrolyte Free Central is available in 3 sizes.2
SmofKabiven Peripheral is available in one size.3
SmofKabiven Central
- SmofKabiven 4 gN, 493 ml
- SmofKabiven 8 gN, 986 ml
- SmofKabiven 12 gN, 1477 ml
- SmofKabiven 16 gN, 1970 ml
SmofKabiven EF Central
- SmofKabiven EF 8 gN, 986 ml
- SmofKabiven EF 12 gN, 1477 ml
- SmofKabiven EF 16 gN, 1970 ml
SmofKabiven Peripheral
- SmofKabiven Peripheral 9.8 gN, 1904 ml
CI = confidence interval; EF = electrolyte free; gAA = grams amino acids; ICU = intensive care unit; N=nitrogen; PN = parenteral nutrition; RR = relative risk.
* This was a systematic review and meta-analysis of 49 randomised, controlled trials (3,641 patients). For the primary endpoint of infection rate, 24 studies were included (2,154 patients), 7 of these were in ICU patients and 17 in non-ICU patients. Thirty-day mortality was a co-primary outcome reported in 20 of the trials.9
** non-ω-3 fatty acid enriched.